How does the developing embryo know where to put the features of the face, the limbs, fingers, organs, bones and all those other amazing facets of our physiology? It has to do with the body’s innate bioelectrical system, says Professor Michael Levin from Tufts University in the US.
In a fascinating experiment, Levin showed that a developing frog embryo generated a series of electrical patterns in the very locations where, a few hours later, the ears, eyes and nose appeared. In other words, the electrical signals generated a blueprint for these features.
One of the ways the body’s electricity is generated is by electrically-charged ions – such as sodium, calcium and potassium. When a cell’s numerous ion channels open, these ions move through, changing the voltage of the cell and creating down-stream effects. It is voltage changes like these that cause the electrical patterns that foreshadow the development of the facial features of a developing tadpole, for example.
Levin describes the process like this. ‘[A]ll cells, not just neurons, have ion-channel proteins and pumps that set their resting potential …; this is now known to be a key parameter regulating cell-level behaviours such as proliferation, differentiation, apoptosis, migration, and directional polarization in a wide range of cells from yeast to human stem cells.’
Levin has shown that changing the body’s bioelectrical pattern can change its physical appearance, too. By artificially changing voltage states, he managed to develop worms with heads at both ends of their bodies, for example.
tissue-and organ-level bioelectric patterns
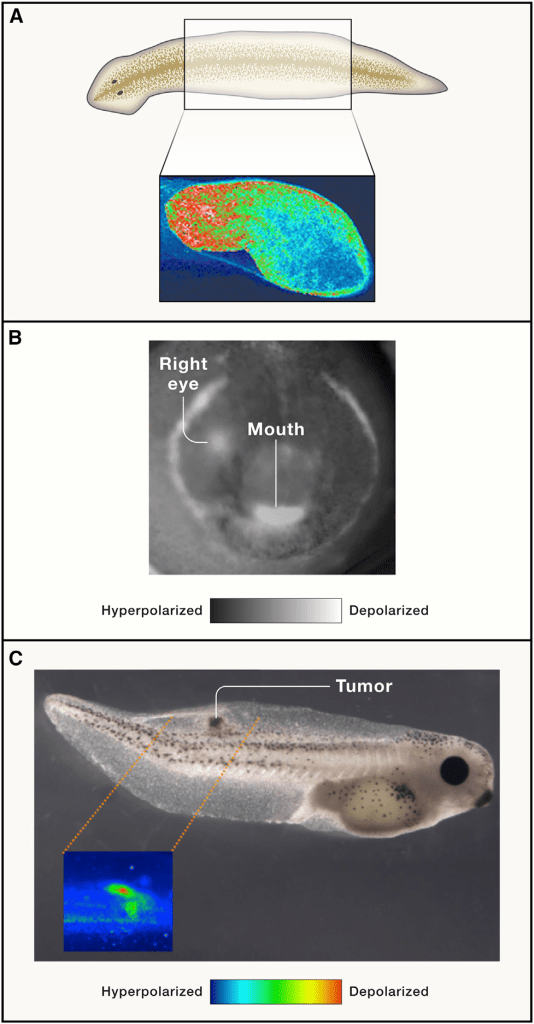
(A) Spatial distributions of Vmem across tissue can be visualized by voltage-sensitive reporter dyes to reveal the depolarized head end and hyperpolarized tail end of the bioelectric circuit in a planarian flatworm fragment that determines its future regenerative anatomical polarity.
(B) Endogenous prepatterns instructively guide morphogenesis, such as this ‘‘bioelectric face’’ distribution on the early frog ectoderm that determines the gene-expression domains and the borders of the compartments for the eyes, mouth, arches, etc. (white signal demarcates depolarized cells, such as the embryo’s right eye, appearing slightly earlier than the left). Drugs, ion-channel misexpression, or optogenetics can be used to alter this pattern resulting in predictable changes in the domains of expression of craniofacial patterning genes and subsequent head anatomy.
(C) Individual cells transforming into cancer depolarize as a very early step in the process, leading to an electrical de-coupling from tissue-level organizational cues (here shown as an early stage of a tumour in tadpoles injected with human oncogenes; voltage-sensitive fluorescent dyes reveal location and size of the cells abandoning their participation in organogenesis in favour of tumorigenesis). **
These findings could have therapeutic applications. Levin says, ‘the ideal bioelectric interventions will permanently re-set patterns towards which cells work. The role of bioelectric circuits in implementing large-scale decision making in cell collectives offers the opportunity to induce repair of disease states (and not simply to address symptoms)…’
Kcnj2 (Kir2.1) potassium channel mutations in mice result in deformations in craniofacial and limb structures, for example. Phenotypes include cleft palate, hypoplastic tympanic ring, nasal bones, maxilla, premaxilla, mandible, and enlarged fontanelle. Limb defects include extra digits and digits that are reduced in size.
Human patients with mutations in kcnj2 (Kir2.1) potassium channels exhibit Andersen-Tawil syndrome, with craniofacial malformations that include a cleft or high arched palate, broad forehead and nose, wide-set eyes (hypertelorism), low-set ears, and a small lower jaw (micrognathia). Individuals with mutations in kcnj2 often also have digit defects such as clinodactyly (abnormal curvature), brachydactyly (shortened digits), and syndactyly (fused digits).
While the developmental bases of these disorders are currently the focus of much research, it is already clear that endogenous non-neural bioelectric signalling via potassium channels and gap junctions is essential for normal development and regeneration.
reprogramming anatomy
The ability to stably derive a different anatomical structure from a wild-type (natural) genome was originally described in the inheritance of cortical surgical interventions in propagating lines of single cell ciliates (Beisson and Sonneborn, 1965). However, recent work has shown that permanent reprogramming of the entire body plan is possible in complex multicellular organisms as well.
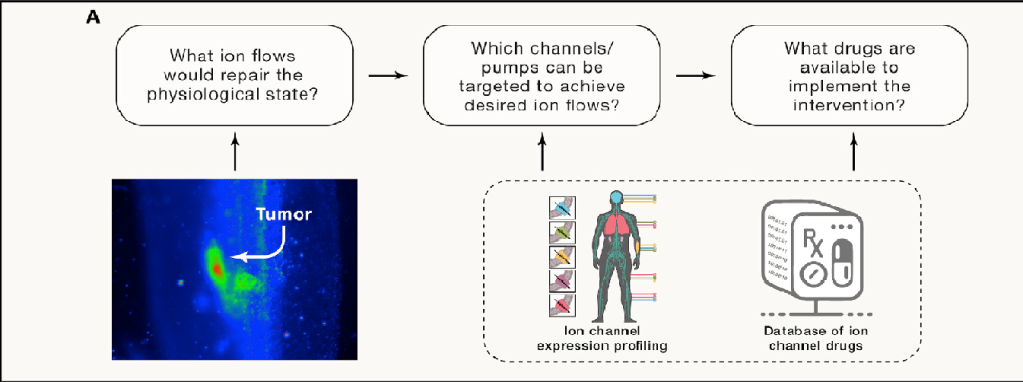
roadmap for electroceutical biomedicine
The large set of known ion-channel drugs, many of which are human approved, can be exploited in regenerative medicine. Existing transcriptomic databases reveal which ion channels are potential targets for intervention in any tissue and serve as inputs to a computational environment that simulates bioelectric circuits and thus can predict the large-scale patterns that would result from opening or closing specific channels. These in silico models can be used to derive candidate interventions—blends of specific small-molecule compounds that target tissue-specific ion channels to induce the desired bioelectric state to trigger repair, remodelling, or normalization, as needed.
Driving growth in bioelectronic medicine is a convergence of advances in neuroscience, electronics, materials science, molecular medicine, and biomedical engineering, alongside more than a billion dollars of investments from government and industry. Within the next decade, researchers say, modulating the body’s neural networks could become a mainstream therapy for many of today’s greatest health issues—from arthritis, asthma, and Alzheimer’s disease to depression, diabetes, and digestive disorders.**
Author of original article by Lyn McLean, EMR Australia 29 April 2023 and edits curated from the original research papers by Neurosoma.
Sally Adee, ‘The amazing ways electricity in your body shapes you and your health’, New Scientist 22 February 2023.
* Figure: (A) by Taisaku Nogi. Images in (B) and (C) are used with permission from Chernet and Levin (2013b) and Vandenberg et al. (2011) from Levin, Bioelectric Signaling: Reprogrammable Circuits Underlying Embryogenesis, Regeneration, and Cancer, Cell (2021), https://doi.org/10.1016/j.cell.2021.02.034;
**Lynne Peeples Authors Info & Affiliations The rise of bioelectric medicine sparks interest among researchers, patients, and industry, December 3, 2019. 116 (49) 24379-2438 https://doi.org/10.1073/pnas.1919040116

